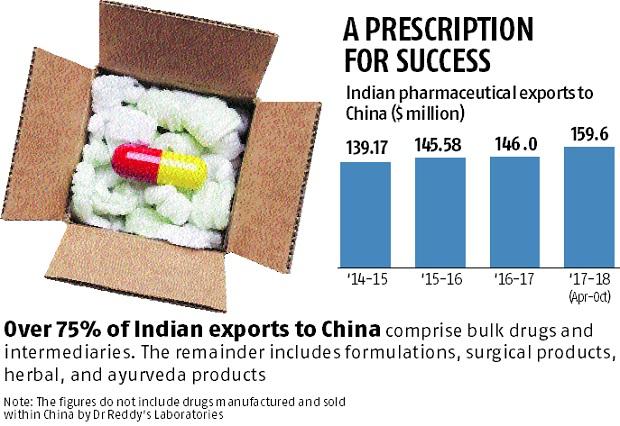
Dr Reddy’s Laboratories (DRL) is expanding its presence in China, the world’s second-largest pharmaceutical market, where other drug manufacturers such as Cipla and Lupin are exploring opportunities.The more than $100-billion China market is dominated by local drug manufacturers and multinationals and India’s pharmaceutical exports of around $160 million are a fraction of the companies’ sales.
But recent changes in regulation allowing quicker product approval and growth opportunities in China have been drivers for Indian companies.According to The Pharma Letter website, China’s Food and Drug Administration recently decided to acknowledge foreign trial data and hire more people to speed up drug approvals — moves that could benefit India’s drug manufacturers. “While Indian companies have traditionally focused on the US and Europe, new regulations in China, which could fast-track approvals for products cleared by the US Food and Drug Administration, could help Indian companies secure a foothold in the market,” said Sriram Shrinivasan, global emerging markets leader, lifesciences, EY.DRL said it was planning to introduce anti-cancer drugs, while Cipla and Wockhardt are planning to launch core therapies such as antibiotics and respiratory drugs in China.
“A strong pipeline is in place and this will continue to be strengthened along with the expansion of our field force. Oncology is an area of focus. We are also looking at partnerships to expand our commercial footprint in the China market,” said DRL Executive Vice-President M V Ramana.DRL manufactures and sells drugs in China in a joint venture with Canada’s Rotam group. The joint venture (JV) has a sales and marketing team covering 5,000 hospitals spread over China.DRL sells products worth around $20 million in China, according to global health care services firm IQVIA. However, the figure does not include sales of active pharmaceutical ingredients.While DRL has been present in the Chinese market for nearly 20 years, other Indian companies have had limited success there.
Ranbaxy, which was acquired by Sun Pharmaceuticals in 2014, exited from a JV in China and sold its stake to its partners in 2009. Torrent dropped its plans to launch drugs in China because of stringent regulatory requirements. In 2013, the firm had initiated talks with a Chinese company but the plan was dropped two years later, a company spokesperson said. Three years ago Cipla exited from two investments it had made in China.But drug manufacturers are preparing to take fresh bets in the market with a focus on core therapies. The operating environment will remain challenging but strong demand and quick approval for innovative products in China are an attraction, according to IQVIA.Wockhardt is planning to launch its antibiotics in China and is in talks with local companies for clinical trials. The company is developing five antibiotics.Cipla is keen to make a foray into the respiratory segment through an acquisition or partnership. A spokesperson for the company was not available for comment. “We will explore China because it is a very large market for antibiotics,” Wockhardt Chairman Habil Khorakiwala stated. The antibiotics market in China is double that of the US by value and is 25 times bigger in volumes, indicating a huge potential for Wockhardt’s novel drug pipeline. “In China we intend to outlicense the antibiotics. We studied the market and found it is too complex for us to market drugs,” Khorakiwala added.Fabrice Egros, Lupin’s president for the Asia-Pacific region, said the company was exploring opportunities for partnerships in China.However, cultural challenges, along with the fragmented nature of the market, have been seen as hindrances.“Indian companies have not focused on China because of complexities and higher opportunities in markets like Europe and Japan, which are the focus areas after the US,” said D G Shah, secretary-general, Indian Pharmaceutical Alliance.
