INTRODUCTION
Nanotechnology is clearly a concept whose time has come. Five years ago, little was known about the technology outside specialist circles. Yet it is now being promoted in the scientific and popular press as a major technological breakthrough, heralding the next industrial revolution. Researchers and developers are talking about how nanotechnology might be used to develop lighter, stronger materials, better batteries and improved solar cells in the near-term (just a few of the many examples), with applications such as targeted cancer treatments, microscopic sensors and even life-mimicking devices in the mid to distant future. This enthusiasm is backed up by serious research and development funding from government and industry—estimated at nearly US$10 billion globally for 2005.
Born from the marriage of nanotechnology and medicine, nanomedicine is set to bring advantages in the fight against unmet diseases. The field is recognized as a global challenge, and countless worldwide research and business initiatives are in place to obtain a significant market position.
MARKET FOR NANOMEDICINE
In the field of nanomedicine research, the US accounts for one-third of all publications and half of patent filings. A comparison between Europe as a whole and the US shows that while Europe is at the forefront of research, the US leads in the number of patent filings. The strong patenting activity of US scientists and companies indicates a more advanced commercialization status.
According to BCC Research (www.bccresearch.com), the global market for nanoparticles in the life sciences is estimated at over $29.6 billion for 2014. This market is forecast to grow to more than $79.8 billion by 2019, to register a healthy compound annual growth rate (CAGR) of 22%. The biggest increase will come in the area of drug delivery systems.
As products complete clinical trials and gain US FDA market approval, the revenues from these products will grow at 23%. Basic biotechnology research revenues will increase due to the quest to find more nanoparticle applications, as more drugs become successfully delivered by these carrier systems. Drug development and formulation will show steady sustained growth at 20.7%. Nanoparticles for use in diagnostic imaging will continue to show healthy growth at 20.1%. This will result from the need to develop more definitive nanoparticle markets for disease diagnosis.
According to a research report from the Business Communications Company (BCC) Research, despite the catastrophic consequences of the 2008-2009 crisis on capital markets, the global nanomedicine sector, which was worth $53 billion in 2009, is projected to grow at a compound annual growth rate (CAGR) of 13.5%, surpassing $100 billion in 2014. One of the largest segments
of this market is represented by anticancer products. Valued about $20 billion in 2009, it is expected to reach $33 billion in 2014, growing at a CAGR of 11%.
Of the approximately 200 companies identified as active in nanomedicine worldwide, some three- quarters are startups and SMEs focusing on the development of nanotechnology-enhanced pharmaceuticals and medical devices.
Nano-enabled medical products began appearing on the market over a decade ago and some have become best-sellers in their therapeutic categories. The main areas in which nanomedical products have made an impact are cancer, CNS diseases, cardiovascular disease, and infection control.
At present, cancer is one of the largest therapeutic areas in which nano-enabled products have made major contributions; these include Abraxane, Depocyt, Oncospar, Doxil, and Neulasta. Cancer is a prime focus for nanopharmaceutical R&D, and companies with clinical-stage developments in this field include Celgene, Access, Camurus, and Cytimmune.
Treatments for CNS disorders including Alzheimer’s disease and stroke also feature prominently in nanotherapeutic research, seeking to build on achievements already posted by products such as Tysabri, Copazone, and Diprivan. According to BCC Research, this is a field hungry for successful therapeutic advances and annual growth from existing and advanced pipeline products is expected to reach 16% over the next 5 years.
Autoimmune-related inflammatory disease has an increasingly high profile, and nanotechnology has contributed to the success of products such as Remicade and Humira. Enzon is among companies vigorously pursuing new product development in this field, and new products are expected to add to the continuing market penetration of existing therapies, contributing to annual growth rates around 15%.
In addition, nanotechnology has contributed to a wide variety of anti-infective products, from PEGylated interferons used in viral disease to nanocrystalline silver used topically in wound infections. Biosanté and NanoBio are among companies actively involved in this field.
The US market is by far the largest in the global nanomedicine market, and is set to continue to dominate the world marketplace, but other national markets are expected to increase their shares over the next 5 years.
LANDSCAPE
In 2007 investment in nanotechnology by VCs was US $702 million, involving 61 deals. 27% went to healthcare and life science, 31% to energy and environment, and 42% to electronics and IT. Two years later, nanotechnology market captured US $792 million from VCs. Of these, the largest share (51%) went to healthcare and life sciences, followed by energy and environment and electronics and IT, with 23% and 17%, respectively. Doubling the funds invested in the healthcare segment in just two years, the VC industry has demonstrated a clear interest in investment opportunities in the nanomedicine field
REGULATORY FRAMEWORK
Not having a very explicit, legal and concise regulatory guidelines couple with long approval procedure and regulations make nanomedicine products different from those of other industries using nanotechnologies with no limitations due to regulatory bodies. As a consequence, the expenditure to bring a nanomedical product to the marketplace is so huge that pharmaceutical and biotechnology industries have no alternative but focus on the blockbusters that can please the stockholders. It is reasonable to presume that nanomaterials are “new for safety evaluations purposes”, and therefore they merit careful oversight by regulatory authorities both before and after entering the marketplace.
COMMERCIALIZED & FUTURE MARKETS
Apart from targeted cancer chemotherapy, nanotechnology is being used more widely in creating a new generation of drug delivery systems. A key factor in its adoption is that nanoscale particles have a greater surface-to-volume ratio than macroparticles. Thus, a drug-bearing nanoparticle can release a drug more quickly and more abundantly than larger particles. This is helpful when the drug poses problems with solubility and absorption, as is the case with a considerable proportion of new drugs.
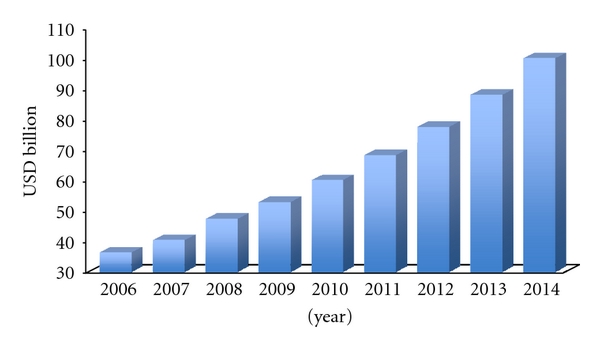
(a) Global Nanomedicine Market Size
Already on the market in the US and elsewhere are wound dressings that exploit the antimicrobial properties of nanocrystalline silver. Ionic silver is a powerful antibacterial, effective even against problem organisms like MRSA, and nanotechnology offers a way to optimize its effect when incorporated in a wound dressing.
Within the anticancer products segment, Doxil and Abraxane are two main examples of success in the clinic. Sequus Pharmaceuticals was the first company to sell doxil, the liposomal formulation of Doxorubicin, a powerful but toxic chemotherapeutic, initially approved for treatment of Kaposi’s sarcoma in the USA in 1995. Sequus was then acquired in 1998 by ALZA Pharmaceutical for US $580 millions, which subsequently merged with Johnson and Johnson in 2001 in a US $12.3 billion deal. The other approved nanotherapeutic agent, Abraxane, instead, was originally sold by Abraxis Biosciences, which was acquired in June 2010 by Celgene Corporation for US $2.9 billions.
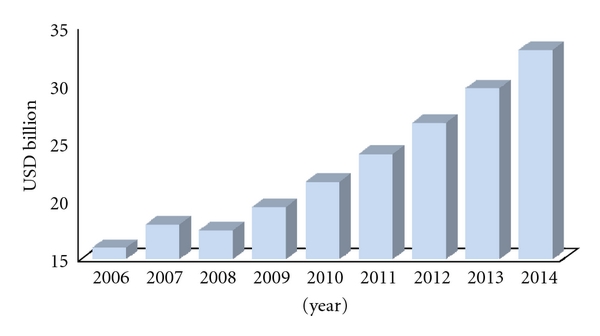
(b) Anticancer Products Market Size
Granted by the orphan drug designation in January 2005 by the FDA, this product consists of albumin nanoparticles containing paclitaxel, and is indicated for the treatment of breast cancer.
Conventional chemotherapies consist of injections of cytotoxic drug intravenously, which indiscriminately kill both healthy and tumor cells. The clinic success of Doxil and Abraxane was driven by their ability to concentrate preferentially in tumors, because of the gaps (otherwise called endothelial fenestrations) characterizing the blood vessels that supply the cancerous mass. Nanoparticles of the right size can penetrate these “gates” and passively diffuse into the tumors. Thanks to this generation of chemotherapies, patients are now benefiting from new treatment strategies for delivering drugs through nanotechnology carriers with lower systemic toxicity and improved therapeutic efficacy
CONCLUSIONS
Despite the issues nanomedicine still has to face, investments in this market are predicted to increase. New applications of nanomedicine have been demonstrated, and the resulting expansion of the potential market makes the risk more appealing. Ferocious financial collapse elevated sunk costs of the essential R&D process, tricky access to funds, uncertainty of expected returns, and the extremely meticulous, and lengthy FDA regulatory process has not deterred the investors’ community. On the other hand, the promises of great future potential developments in the different market segments and high returns connected to the high risk of the innovation investments make this market still considerably attractive. Compared to the 2007 benchmark, VCs in 2009 decided to double their investments in this sector, at the expenses of the information technology market. The fact that nanomedicine dominates the VC funding in the healthcare market is surely a good predictor of the bright future landscape of expansion of this promising area of research.
ABOUT AUTHOR
Tarun Pandotra is Vision-driven executive with 20 years of demonstrated leadership in the pharmaceuticals and CRO industry. Proven track record of driving bold initiatives and innovative partnerships across multiple channels, expanding business scope, productivity, and profitability. Extensive international experience, with commitment to delivering quality results, establishing ethical guidelines, and motivating staff for peak performance and program success. Tarun has authored various articles which was published in Biospectrum Asia, Pharmaphorum and so on.
