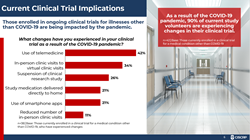
Patients currently enrolled in ongoing clinical trials for medical conditions other than COVID-19 report experiencing changes as a result of the pandemic such as telemedicine, study medication delivered to their homes, and the use of smartphone apps.
BOSTON (PRWEB) MAY 26, 2020
The recent public healthcare crisis has generated an increasing amount of attention on the topic of clinical research. In light of this, CISCRP conducted a brief survey among the public and patients in April 2020 to assess the impact of the pandemic on clinical research perceptions and experiences. The final report can be accessed here.
Five hundred diverse individuals in the United States and a few select countries in Europe (United Kingdom, France, Germany and Italy) completed the survey. Eighteen percent of the respondents have clinical trial experience. Of those who have participated in clinical research, more than half (56%) are currently enrolled in a clinical trial for COVID-19. Where appropriate, comparisons were made to the baseline 2019 Perceptions & Insights Study, a biennial large-scale global analysis of public and patient views of and experiences with clinical research to identify any notable differences.
Overall, awareness of clinical trials remains low – as 58% had not heard of a COVID-19 clinical research study recently.
“Interestingly, among the minority that had recently heard of a COVID-19 clinical research study in the recruitment phase, we learned that a higher proportion (40%) of Europeans are aware than Americans (34%),” said Annick de Bruin, Director, Research Services. “This contrasts with our larger baseline 2019 Perceptions & Insights Study where we found that North Americans are more likely to be aware of a current clinical trial in general than any other region. This may partially be due to the fact that the crisis evolved earlier in Europe.”
Misconceptions about the clinical research development process persist. Most (60%) think a treatment or vaccine for the virus will be developed in less than one year. Once developed, the majority (64%) think it will be less than a year before people can start receiving it.
Patients currently enrolled in ongoing clinical trials for conditions other than COVID-19 report experiencing changes as a result of the pandemic such as telemedicine, study medication delivered to their homes, and the use of smartphone apps. Twenty-six percent report a suspension of the clinical research study in which they were enrolled and 11% report a reduced number of in-person clinic visits.
Top motivators for participating in a COVID-19 clinical research study include altruistic reasons with 46% mentioning ‘to help others who are suffering’ and 46% mentioning ‘to advance science and the treatment of COVID-19.’ These motivations are in line with findings from the larger baseline 2019 Perceptions & Insights Study.
Both the COVID-19 special report, as well as the 2019 baseline reports are available free of charge on the CISCRP website under Research Services, https://www.ciscrp.org/services/research-services/ . CISCRP also presents the aggregate results of the Perceptions and Insights Study at industry conferences, company meetings, and other virtual and in-person forums. For a detailed, in-depth report with a facilitated virtual meeting or webinar, please contact Annick de Bruin at adebruin@ciscrp.org or call 617-725-2750 x400.
About CISCRP
The Center for Information and Study on Clinical Research Participation (CISCRP) is an internationally recognized non-profit organization dedicated to educating and informing the public and patients about clinical research. CISCRP works to raise awareness, enhance experiences, and strengthen communication and relationships among participants, research professionals and the public through various services and events. http://www.ciscrp.org
