
Is this a right time to expand in India?
What is the right business model to be followed?
What is the right geography to start with?
In the Fiscal Tear (FY) 2014-15, India’s GDP grew by 7.3 percent following a 6.9 percent growth rate in the previous year. With the growth of 7 percent in first half FY 2015-16, India is considered the world’s fastest-growing large economy. The International Monetary Fund (IMF) predicted that India would retain the status of fastest growing economy until 2020. Domestic consumption and investment were major contributors to growth in FY2014-15, accounting for 57 per cent and 33.1 per cent of GDP, respectively.
Government of India has introduced key reforms to the FDI policy, to help attract further invest-ments. To achieve this goal, some measures such as the introduction of the composite cap that does away with the distinction between FDI and Foreign Portfolio Investment (FPI) and liberalising FDI norms in 15 major sectors have been taken. FDI in India has started picking up, which stood at USD16.63 billion in 1H FY2015-16, about 13 per cent higher than 14.69 billion in 1H FY2014-1510.
The Government is keen to work with Pharma industry and make suitable amendments in policies to enhance manufacturing activity, inclusion of the pharmaceuticals sector in the “Make in India” campaign has provided a unique opportunity to the industry to push its agenda of R&D, innovation and affordable healthcare for all.
We have a team of specialists those who have experience in developing strategies, be it building market entry strategies, setting up of enhanced distributor network, opening up of a sales office, getting regulatory approvals, managing compliances, joint venture support or advisory services for future growth, we work closely with companies and assist them in taking a lead in this competitive environment.




Regulatory Intelligence and Strategy
With our expertise, collaborations and strong network of industry and regulatory contacts, we pro-vide insightful analysis of Indian regulatory scenario for Pharmaceutical, Medical Device, Diagnos-tic Kits, Biologicals, Borderline products and Vaccine industry.
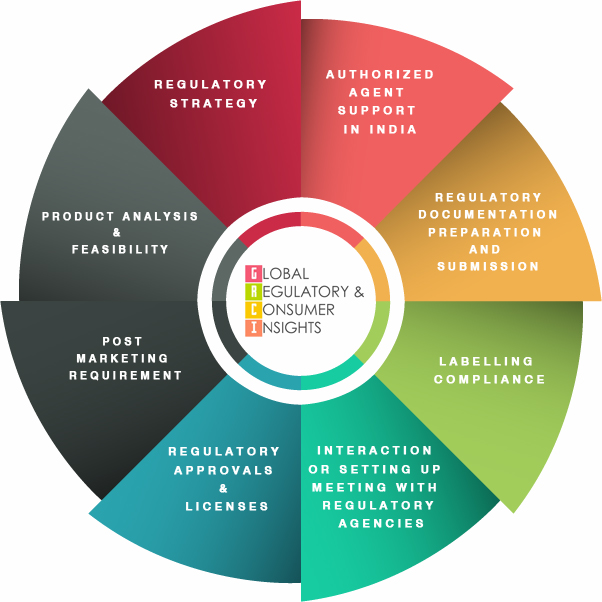
Product Analysis & Feasibility:
During product analysis and feasibility our expert looks into every aspect i.e.
- ♦ Technical
- ♦ Legal
- ♦ Operational
- ♦ Timelines
Regulatory Strategy:
The regulatory scenario of the country have influence on the approval of the product. It is therefore necessary to be aware of the new regulations or updates. Based upon the product analysis, feasi-bility and our strong roots in the industry and professional guidance provides you with the know-how of the updated guidelines and regulations facilitating the filing of appropriate documents for hassle free approvals.
Authorized Agent Support in India:
We can act as authorized agent/registration holder on your behalf for speedy import and market entry of your products.
Regulatory Documentation preparation and Submission:
Dossier preparation with support of checklist for different product as per the guidelines are manda-tory to be followed for successful submissions. We provide assistance in compiling the documents and its validation as per guidelines. Our expert liaise with agency and ensure the timely submission of dossier to the agency. Our experts also assist in timely query responses for your products and filing the appropriate quality documents reducing the chances of rejection.
Labeling Compliance:
With our team of experts the slightest details and updates as per the regulations are monitored and communicated to the client for labeling compliance minimizing the chances of queries.
Labeling Compliance:
With our team of experts the slightest details and updates as per the regulations are monitored and communicated to the client for labelling compliance minimizing the chances of queries.
Interaction or Setting up meeting with Regulatory Agency:
On request from you, we can set up the meeting with Regulatory agency. We also support to schedule the meeting for presenting the protocols to NDAC member. We help our clients through-out the process of regulatory procedures providing step by step support for successful approvals.
Interaction or Setting up meeting with Regulatory Agency:
We support you till the final outcome of your application and ensures that you receive approvals or appropriate license.
Post Marketing Requirements:
Any post marketing changes on the product are notified to the regulatory authorities as per the formats applicable on behalf of our clients. We are in collaboration with Pharmacovigilance com-panies and can help the clients to monitor the effects of medical drugs in post approval marketing. We also review the Periodic Safety Update Report (PSUR) and Adverse Drug Reaction (ADR) reports for our clients enabling them to submit the data to regulatory authorities in India.
Authorise Agent/Registration holder support
The companies with no local presence trying to venture in India need to appoint an Authorise Agent/Registration holder. We with a valid license serve as an Authorise Agent/Registration holder and can be your in-country representative for end-to-end purposes including the liaison with regulatory agency
Distribution Identification
We support our clients to identify distributors to succeed in the Indian market for successful launch of their products.

Network of distributors is an essential part of the company’s supplier ecosystem. Our in depth knowledge of the market and excellent networking with the distributors across India, helps you to provide the mix of distributors.
Consult us to examine your needs, answer your questions and discuss how Global Regulatory & Consumer Insights can best support your project success
For a more detailed discussion on how we can help you, contact us on +91 886000 9879 or email us via our contact form
